ImmunityBio (IBRX)·Q4 2025 Earnings Summary
ImmunityBio Reports 700% Revenue Growth, Wins First Lung Cancer Approval, Stock Hits 52-Week High
February 23, 2026 · by Fintool AI Agent

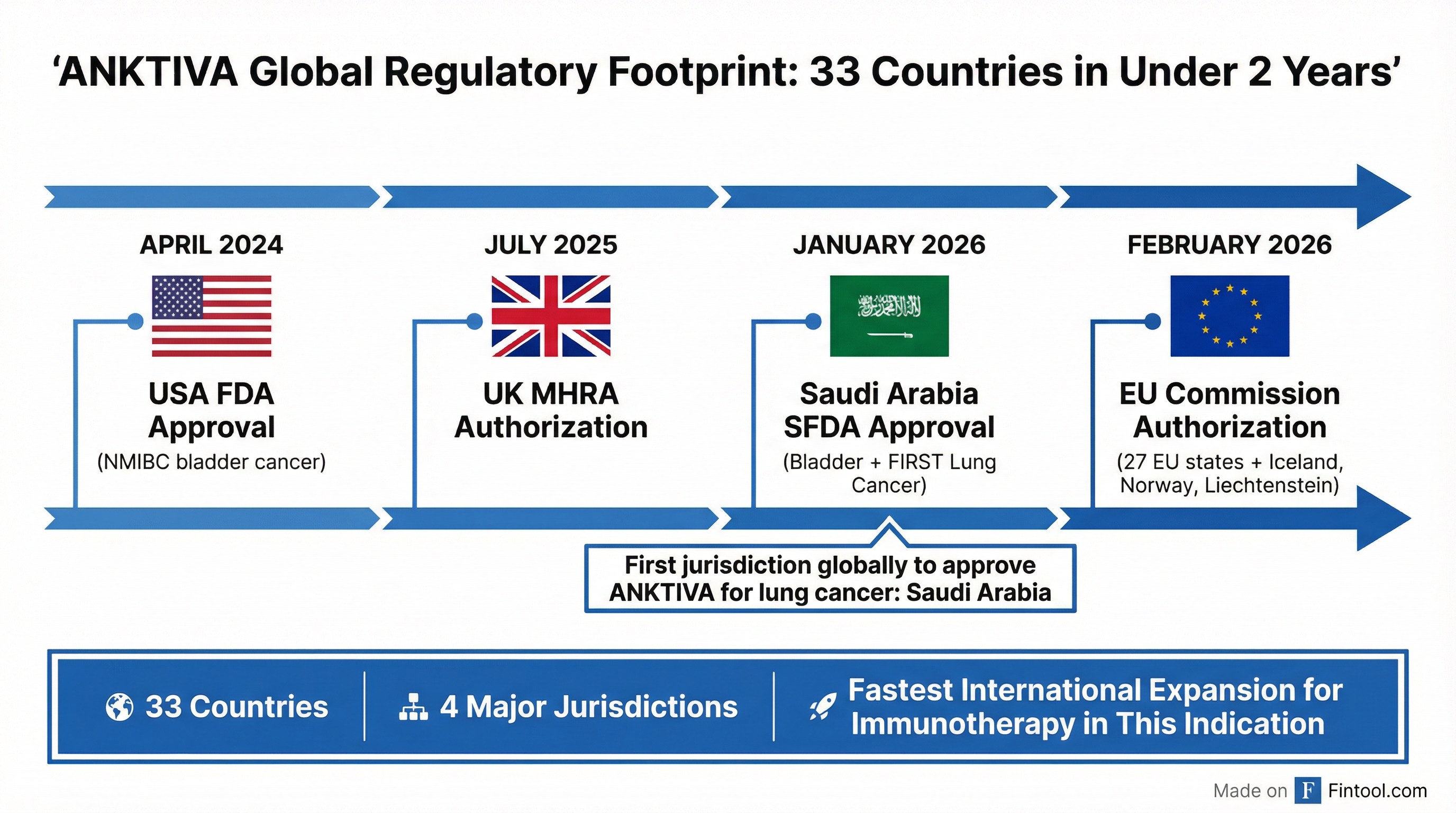
ImmunityBio (NASDAQ: IBRX) delivered a blowout Q4 2025, reporting $38.3 million in ANKTIVA net product revenue—a 431% increase year-over-year—and capping a transformative year with $113 million in full-year product revenue, up approximately 700% from 2024 . The results came alongside a string of regulatory wins that expanded the company's global footprint to 33 countries and secured the first-ever approval for ANKTIVA in lung cancer.
Did ImmunityBio Beat Earnings?
Yes—handily on both revenue and EPS.
ANKTIVA unit sales volume surged 750% year-over-year, reflecting accelerating clinical adoption in bladder cancer . The company also reported a 20% quarter-over-quarter increase in net product revenue, demonstrating sustained commercial momentum heading into 2026 .
What Drove the Quarter?
Three catalysts converged to make Q4 2025 a watershed moment:
1. European Union Authorization
The European Commission granted conditional marketing authorization for ANKTIVA in February 2026, covering all 27 EU member states plus Iceland, Norway, and Liechtenstein . This triggered the +41% single-day stock move on February 18.
2. First Lung Cancer Approval Globally
Saudi Arabia's SFDA granted conditional accelerated approval for ANKTIVA in combination with checkpoint inhibitors for metastatic non-small cell lung cancer (NSCLC) in January 2026—making it the first jurisdiction globally to authorize ANKTIVA outside of bladder cancer . Commercial launch is planned within 60 days.
3. Commercial Infrastructure Build-Out
ImmunityBio established strategic partnerships to support global expansion:
- Europe: Partnered with Accord Healthcare deploying 100+ commercial professionals across 30 countries; established Dublin subsidiary
- Middle East: Partnered with BioPharma & Cigalah for Saudi Arabia and MENA region; formed KSA subsidiary
How Did the Stock React?
IBRX had a spectacular week, driven by the EU approval news and earnings confirmation:
The stock hit a 52-week high of $9.25 on February 20, up from a 52-week low of $1.83. Current price: $8.70 with a market cap of ~$8.6 billion.
What Did Management Say?
Dr. Patrick Soon-Shiong (Founder, Executive Chairman, Global Chief Scientific and Medical Officer):
"In under two years from initial FDA approval, ImmunityBio has built a global commercial footprint spanning 33 countries across four regulatory jurisdictions, with $113 million in full-year net product revenue representing 700% year-over-year growth. The SFDA's accelerated approval of ANKTIVA in combination with checkpoint inhibitors for metastatic NSCLC marks a defining moment for the Company—the first authorization of ANKTIVA beyond bladder cancer and the first validation of its role as a lymphocyte-stimulating agent in solid tumors."
Richard Adcock (President and CEO):
"Our 2025 financial results reflect the growing clinical adoption of ANKTIVA as a foundational backbone of immunotherapy for bladder cancer. With subsidiaries now established in Dublin and the Kingdom of Saudi Arabia, we have the infrastructure to support sustained commercial growth across all 33 countries where ANKTIVA is authorized."
What Changed From Last Quarter?
*Estimated based on prior quarter trends
The most significant change: ANKTIVA's addressable market expanded dramatically from bladder cancer only to now include NSCLC, with label expansion plans underway globally .
Full-Year 2025 Financial Summary
*Calculated from 750% unit growth
SG&A expense decreased $18.8 million driven by lower litigation settlements and commercial consulting costs, while R&D increased due to a $14 million one-time fixed asset write-off and higher clinical trial costs .
What's the Pipeline Outlook?
ImmunityBio outlined a 3-year global strategy positioning ANKTIVA as the backbone of its "Cancer BioShield Platform" across multiple indications:
Near-Term Catalysts (2026)
Active/Planned Trials
Over 30 active and planned clinical trials across 10 tumor types :
- Bladder Cancer: BCG-naïve randomized trial, papillary-only, rBCG shortage solution
- Lung Cancer: 1L and 2L NSCLC combinations
- Pancreatic Cancer: ANKTIVA + CAR-NK (RMAT Designation)
- Glioblastoma: Completed enrollment, randomized trials planned
- Non-Hodgkin Lymphoma: CAR-NK combinations
- Sepsis/Lymphopenia: Phase 2/3 trials in progress
Risks and Considerations
-
Profitability: Despite 700% revenue growth, the company remains deeply unprofitable with $351M net loss in 2025
-
Cash Runway: $242.8M cash vs ~$305M annual operating cash burn suggests ~9-10 months runway without additional financing
-
Regulatory Risk: US FDA pathway for lung cancer approval still uncertain; company planning discussions in 2026
-
Execution Risk: Commercial expansion to 33 countries requires significant infrastructure investment
-
Competitive Landscape: Checkpoint inhibitor market is highly competitive; ANKTIVA must demonstrate differentiated value
-
Litigation: Pomerantz Law Firm investigating claims on behalf of investors (multiple press releases in February 2026)
Key Takeaways
- Revenue momentum accelerating: 20% QoQ growth, 700% YoY growth
- Global expansion achieved: 33 countries in under 2 years from first approval
- Platform validation: First lung cancer approval opens massive new market
- Stock at 52-week highs: +375% from 52-week low of $1.83
- Path to profitability unclear: Still burning ~$300M+ annually
Data sourced from ImmunityBio 8-K filed February 23, 2026, and S&P Global.